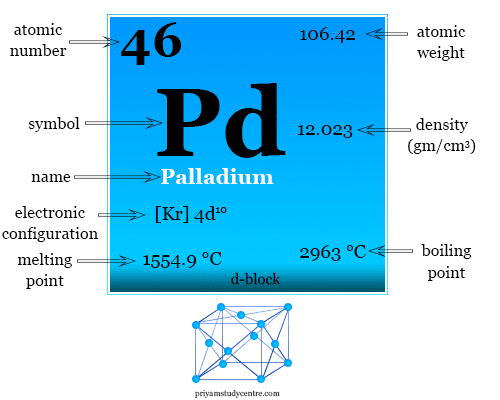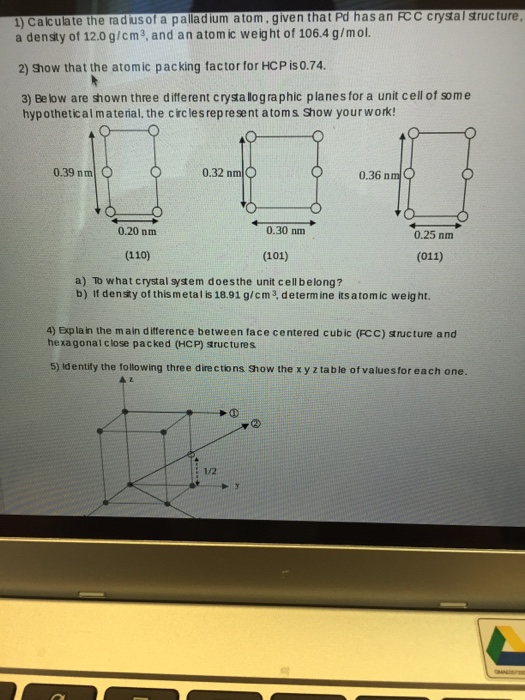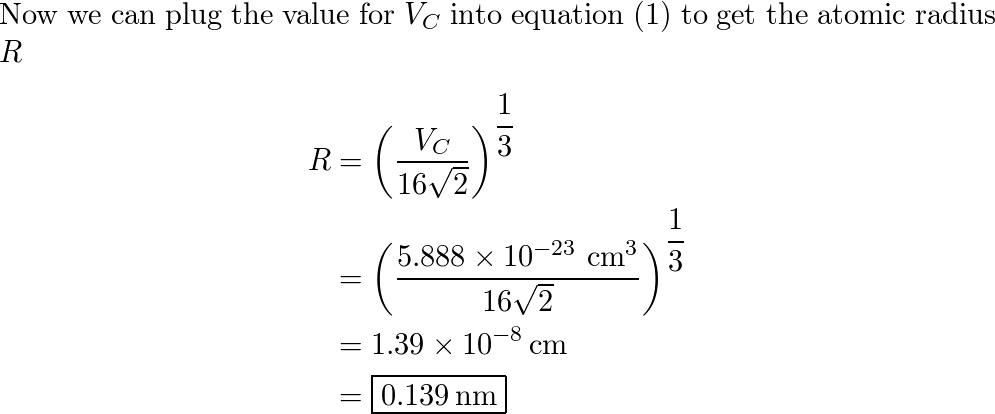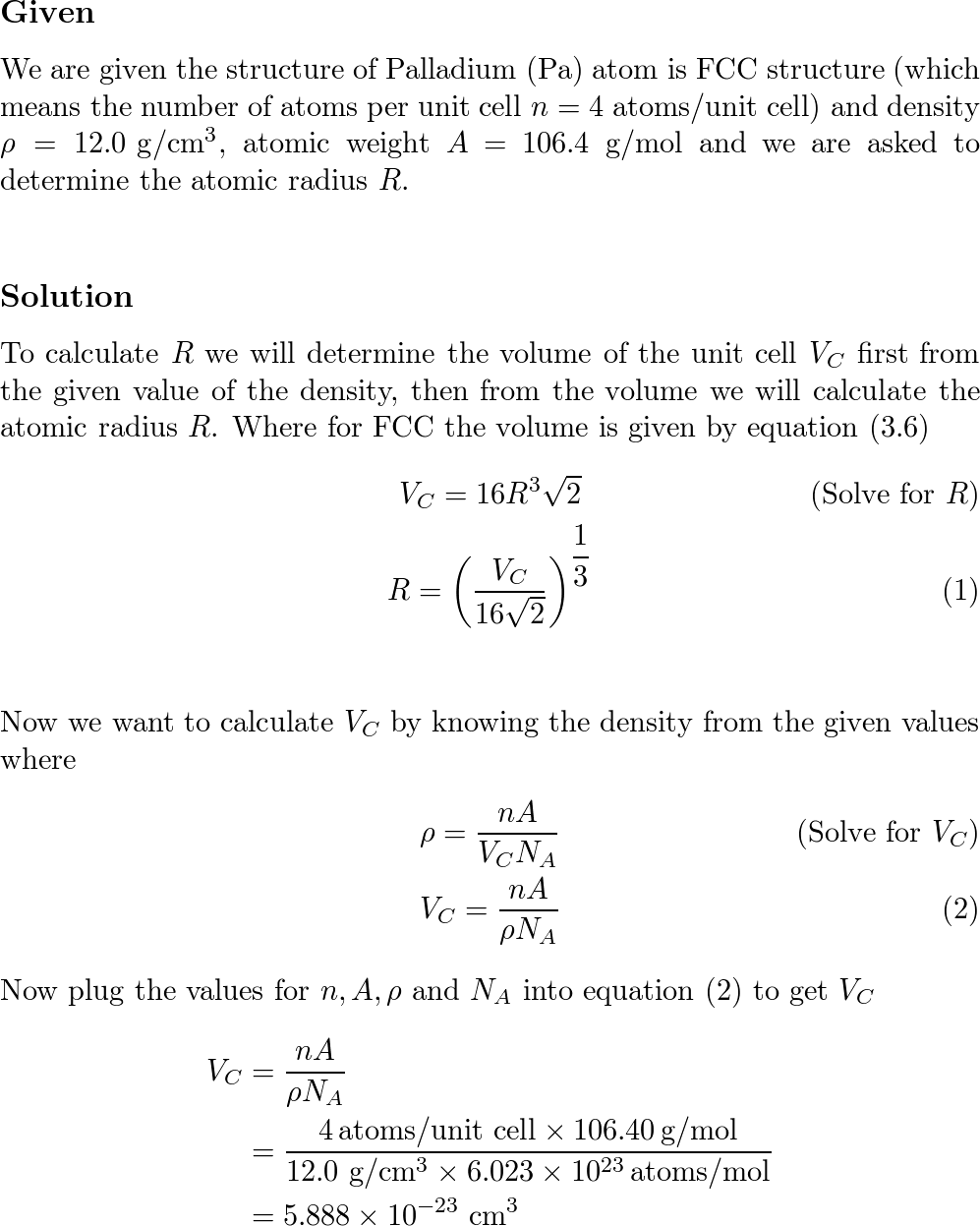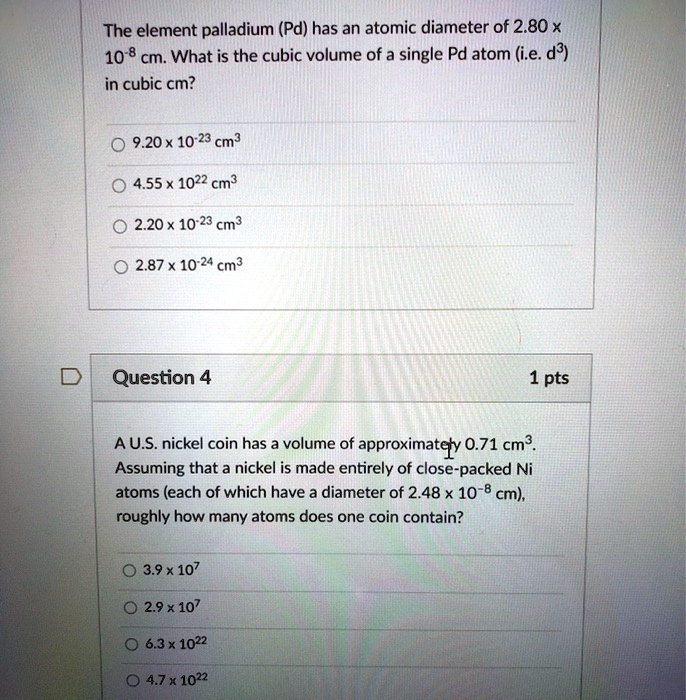
SOLVED: The element palladium (Pd) has an atomic diameter of 2.80 x 10-8 cm: What is the cubic volume of a single Pd atom (i.e: d3) in cubic cm? 9.20x 10-23 cm3
ME 212 Materials Science 11 March 2016 Chapter 6 Problem Set 6.2 Calculate the number of vacancies per cubic meter in iron at 85

Module-10.pdf - Numerical on vacancy defects The density of a sample of FCC palladium is 11.98 g/cm3 and its lattice parameter is 3.8902 Å. Atomic | Course Hero

Magnetic silica surface functionalized palladium catalyst: A modular approach for C-C cross-coupling reactions in water - ScienceDirect

Palladium Acetate Density: 2.19 G/Cm Gram Per Cubic Meter (G/M3) at Best Price in Mumbai | Indian Platinum Private Limited

High Purity Metal Palladium, Electrochemical Electrode, Palladium Electrode, Pure Palladium Wire, Purity 99.95+, Diameter 0.6mm, - Ph Meters - AliExpress